|
Since losing 2 electrons needs more energy than losing one, the alkaline. Despite having some similarities in their physical properties, they are placed in different groups mainly because the alkaline earth metals have two electrons in the outermost shell of their atom. Urbansky ET (2002) Fate of fluorosilicate drinking water additives. Alkaline earth metals are the Group 2 elements in the periodic table. 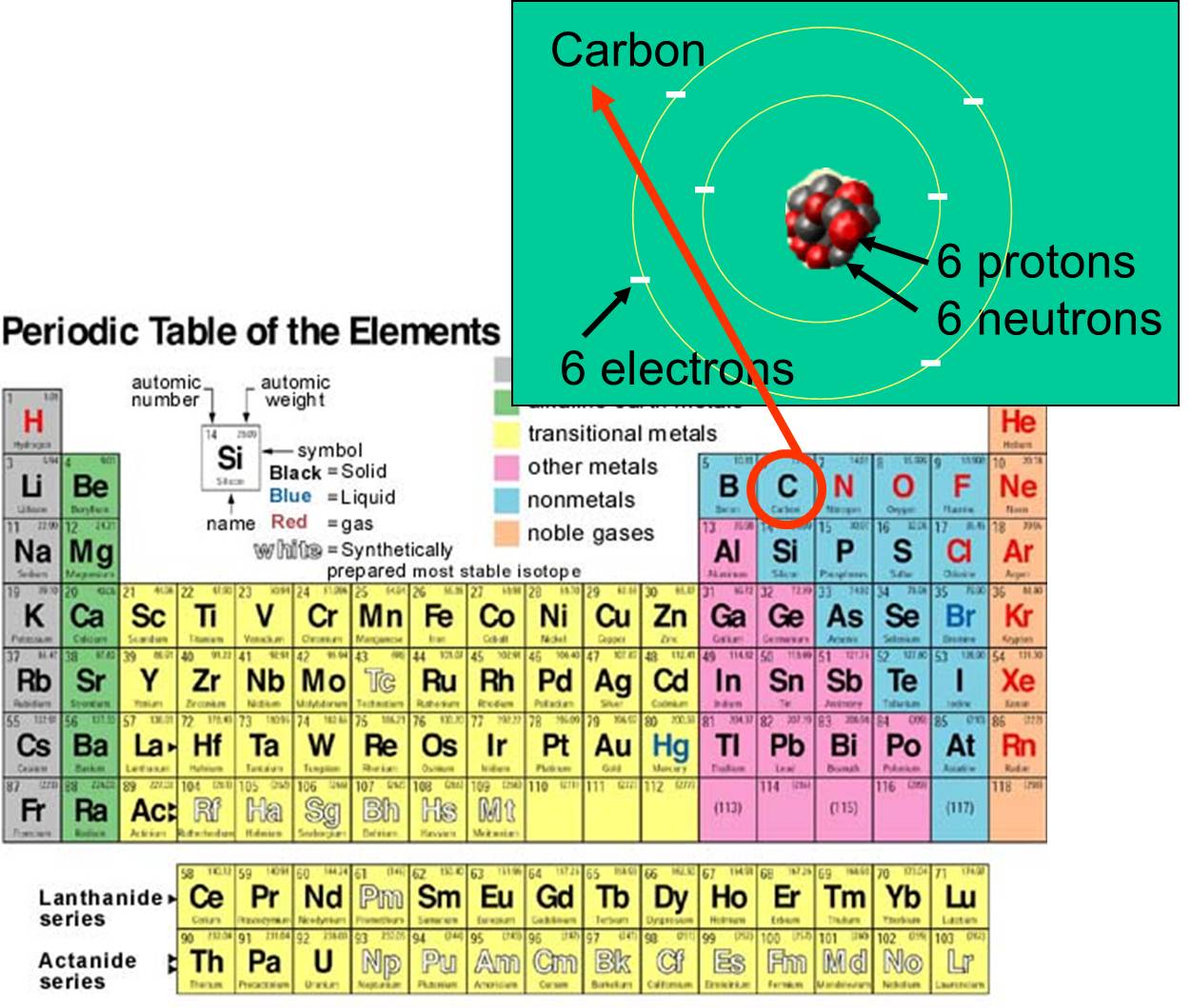
Spivack AJ, You C-F, Smith HJ (1993) Foraminiferal boron isotope ratios as a proxy for surface ocean pH over the past 21-myr. Sanyal A, Hemming NG, Hanson GN, Broecker WS (1995) Evidence for a higher pH in the glacial ocean from boron isotopes in Foraminifera. Rustad JR, Bylaska EJ, Jackson VE, Dixon DA (2010) Calculation of boron-isotope fractionation between B(OH) 3(aq) and B(OH) 4 −(aq). Olah GA, Prakash S, Molnar A, Sommer J (2009) Superacid chemistry, 2nd edn. group: A vertical column in the periodic table. Jagtap SM, Yenkie K, Labhsetwar N, Rayalu S (2012) Fluoride in drinking water and defluoridation of water. atomic number: The number, equal to the number of protons in an atom, that determines its chemical properties. Hemming NG, Hanson GN (1992) Boron isotopic composition and concentration in modern marine carbonates. /periodic-table-165930186-590f2d703df78c92832fe141.jpg)
J Fluor Chem 101:151–153ĭagani R (2003) Feast for Fluorine Aficionados. By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (18341907) in 1869, the table places elements into columnsgroupsand rowsperiodsthat share certain properties. 
A material that conducts heat or electricity, which means. They agree that a transition metal is an element that forms at least one stable ion with a partially filled d-subshell of electrons, but they also say that transition metals can be elements whose atoms have a partially filled d-subshell. A chemical symbol is a notation of one or two letters representing a chemical element. Tungsten has an extraordinarily high melting point, and it's no accident that this element is widely used in light bulb filaments.Christe KO, Dixon DA, McLemore DK, Wilson WW, Sheehy J, Boatz JA (2000) On a quantitative scale for Lewis acidity and recent progress in polynitrogen chemistry. IUPAC (the International Union of Pure and Applied Chemistry) actually has a slightly different definition for a transition metal. Finally, metals are usually dense (that is they have a high mass per unit volume), and they have high boiling and melting points. Their conductivity is a consequence of having electrons that are not tightly bound to the nuclei. They are ductile, which is a fancy way of saying that metals can be made into wires this property convenient because most metals are good conductors of electricity and heat, making them crucial to modern industrial applications. They are typically hard, although potassium and sodium, which serve as biologically active ions in the human bloodstream, can be cut with an ordinary knife. They are malleable, meaning that they can be physically shaped into thin sheets without fracturing. 
They have luster, meaning that they reflect light, a property that often affords them value (e.g., copper, silver). Metals are solid at room temperature with the notable exception of mercury, a liquid used in older thermometers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
